 The strongest lines in the hydrogen spectrum are in the far UV Lyman series starting at 124 nm and below. These images show (a) hydrogen gas, which is atomized to hydrogen atoms in the discharge tube (b) helium and (c) mercury. It is defined as A-logT, where T is transmittance. In this way, we now know the chemical makeup of not just any star, but even galaxies of stars so distant that their light started on its way to us long before Earth had even formed.īohr’s model of the hydrogen atom was a great step forward in our understanding of the atom. 3: The Emission Spectra of Elements Compared with Hydrogen. Absorbance of light is used for most UV-Vis spectroscopy applications. TThe wavelengths of light that are absorbed depends on what elements and compounds it is made of. Absorption Spectrum: When starlight passes through a cloud of gas, some of the light is absorbed and some is transmitted through the gas. Then they can use this knowledge to identify the elements in celestial bodies. The broad range of colors that a star emits depends on its temperature. This means that each type of atom shows its own unique set of spectral lines, produced by electrons moving between its unique set of orbits.Īstronomers and physicists have worked hard to learn the lines that go with each element by studying the way atoms absorb and emit light in laboratories here on Earth. For our purposes, the key conclusion is this: each type of atom has its own unique pattern of electron orbits, and no two sets of orbits are exactly alike. However, because these other atoms ordinarily have more than one electron each, the orbits of their electrons are much more complicated, and the spectra are more complex as well. A bright line, or emission spectrum, appears as a pattern or series of bright lines it consists of light in which only certain discrete wavelengths are present. Similar pictures can be drawn for atoms other than hydrogen. A dark line, or absorption spectrum, consists of a series or pattern of dark linesmissing colorssuperimposed upon the continuous spectrum of a source. By absorbing energy, the electron can move to energy levels farther from the nucleus (and even escape if enough energy is absorbed). Spectrum, in physics, the intensity of light as it varies with wavelength or frequency. In 1814, the German physicist Joseph Fraunhofer observed that the spectrum of the Sun shows dark lines crossing a continuous band of colors. 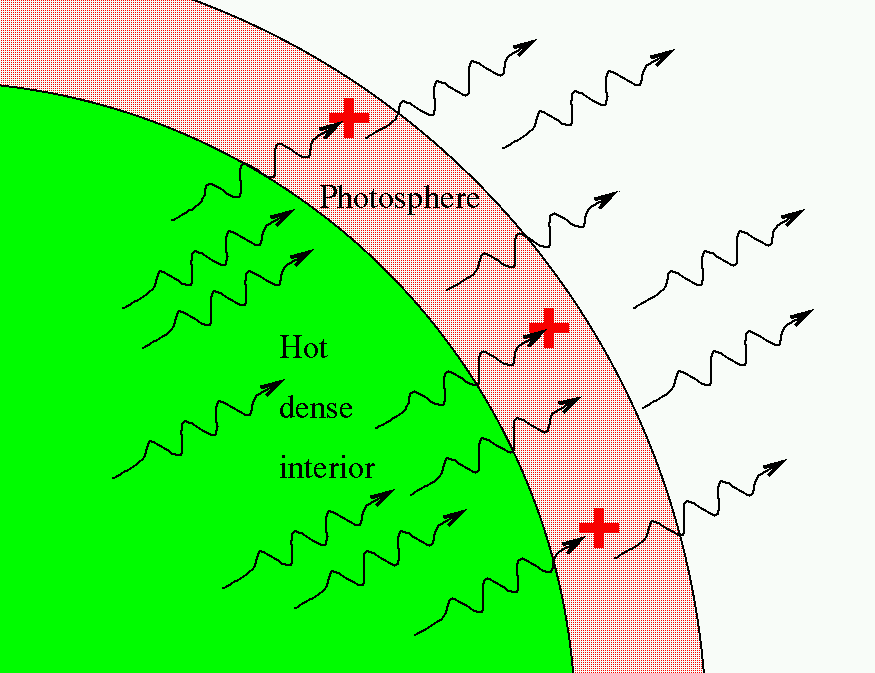
Another way is to use a spectrograph to spread out the light into a spectrum (see the Radiation and Spectra and the Astronomical Instruments chapters). The light spectrum is the consequence of electron transition between different energy levels of orbital for an element. The closer the electron is to the nucleus, the more tightly bound the electron is to the nucleus. Measuring colors is only one way of analyzing starlight. An electron in a hydrogen atom can only exist in one of these energy levels (or states). 
In this simplified model of a hydrogen atom, the concentric circles shown represent permitted orbits or energy levels. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
